Q1. What are the three steps in the halogenation reaction?
Solution
Three steps in halogenation reaction are initiation, propagation and termination.
Q2. IUPAC name of (CH3)3C – CH = CH2 is:
Solution
IUPAC name of (CH3)3C – CH = CH2 is 3,3-dimethylbut- 1 -ene.
Q3. What is meant by inductive effect?
Solution
Polarisation of 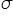 -bond caused by the polarisation of adjacent
-bond caused by the polarisation of adjacent 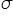 -bond is referred to as the inductive effects.
-bond is referred to as the inductive effects.
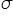 -bond caused by the polarisation of adjacent
-bond caused by the polarisation of adjacent 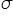 -bond is referred to as the inductive effects.
-bond is referred to as the inductive effects.
Q4. What is the order of rate of reaction of alkanes with halogens F2, Cl2, Br2 and I2?
Solution
The order of rate of reaction of alkanes with halogens is F2 > Cl2 > Br2 > I2.
Q5. Which type of hybridisation of each carbon is there in the following compound?
CH3 – CH = CH – CN
Solution
The hybridisation of each carbon in the given CH3 – CH = CH – CN compound is sp3, sp2, sp2, sp.
Q6. What is the electrophilic centre in the molecule CH3CN?
Solution
In the molecule CH3CN, the starred carbon atom is the electrophilic centre as has partial positive charge due to polarity of bond.

Q7. Write the different possible types of stereoisomers.
Solution
Different possible types of stereoisomers are geometrical isomers and optical isomers.
Q8. What is fractional distillation process?
Solution
When the difference in boiling points of two liquids is not much, simple distillation cannot be used to separate them. The vapours of such liquids are formed within the same temperature range and are condensed simultaneously. The technique of fractional distillation is used in such a process.
Q9. What are polar reactions?
Solution
Organic reactions which proceeds through heterolytic bond cleavage are called polar reactions.
Q10. How can be covalent bonds cleaved?
Solution
Covalent bonds can be cleaved by homolytic and heterolytic cleavage.
Q11. How can you separate aniline from aniline-water mixture?
Solution
Steam distillation technique is applied to separate substances which are steam volatile and are immiscible with water like aniline from aniline-water mixture.
Q12. Write the structure of diphenylamine.
Solution
Structure of diphenylamine is C6H5NHC6H5.
Q13. What is stereoisomerism?
Solution
Compounds having the same constitution and sequence of covalent bonds but differ in relative position of their atoms or groups in space are called as stereoisomerism.
Q14. Which technique one can employ to separate crude oil in petroleum industry?
Solution
Fractional distillation technique can be used to separate crude oil in petroleum industry.
Q15. Draw the structure of 3-Nitrocyclohexene.
Solution
The structure of 3-Nitrocyclohexene is as follows:

Q16. What is geometrical isomerism?
Solution
When the same molecular formula represents two compounds which differ in the spatial arrangement of atoms or groups around C-C double bond, such isomers are called as geometrical isomers. Geometrical isomerim is due to the restricted or hindered rotation around the double bond.
Q17. The correct representation of 4-hydroxy-2-methylpent-2-en-1-al is:
Solution
The correct representation of 4-hydroxy-2-methylpent-2-en-1-al is:

Q18. What are the different types of organic reactions?
Solution
Different types of organic reactions are: Substitution, addition, elimination and rearrangement reactions.
Q19. What is Rf value?
Solution
Distance moved by the substance from the base line to the distance moved by the solvent from the base line is known as the Rf value.
Q20. What is partition chromatography?
Solution
Partition chromatography is based on continuous differential partitioning of components of a mixture between stationary and mobile phases. Example is paper chromatography.
Q21. What is a homolytic cleavage?
Solution
In hemolytic cleavage one of the electrons of the shared pair in a covalent bond goes with each of the bonded atoms. Thus in this movement of a single electron takes place instead of an electron pair.
Q22. Categorise the following ions as nucleophile or electrophile? HS-, BF3, NO2+, C2H5O-, NH2-
Solution
Nucleophile among the following are : HS-, C2H5O-, NH2- Electrophile among the following are : BF3, NO2+
Q23. What is isomerism?
Solution
The phenomenon of existence of two or more compounds possessing the same molecular formulae but different properties is known as isomerism.
Q24. What is heterolytic cleavage?
Solution
In the heterolytic cleavage the bond breaks in a way that the shared pair of electrons remains with one of the fragments.
Q25. Draw the structure of Chlorocyclohexane.
Solution
The structure of Chlorocyclohexane is as follows:

Q26. What is a nucleophile and what is an electrophile?
Solution
A reagent that brings an electron pair is known as nucleophile, and a reagent that takes away an electron pair is called as Electrophile.
Q27. What are the common techniques used for purification of organic compounds?
Solution
The common techniques used for purification are: Sublimation, Crystallisation, Distillation, Differential extraction and Chromatography.
Q28. How to estimate sulphur in an organic compound?
Solution
A known mass of an organic compound is heated in a Carius tube with sodium peroxide or fuming nitric acid. Sulphur present in the compound is oxidized to sulphuric acid. It is precipitated as barium sulphate by adding excess of barium chloride solution in water. The percentage of sulphur can be estimated from the mass of barium sulphate.
Q29. How many σ and π bonds are present in HC ≡ C – CH = CH – CH3?
Solution
In HC ≡ C – CH = CH – CH3, there are 10σ and 3π.
Q30. What are the methods to estimate nitrogen?
Solution
Methods to estimate nitrogen are Dumas Method and Kjeldahls Method.
Q31. What types of reactions alkynes undergo?
Solution
Majority of the reactions of alkynes are the examples of addition reaction.
Q32. What is a substitution reaction? Give an example.
Solution
When one or more hydrogen atoms of alkanes are replaced by halogens, nitro group or sulphonic acid group it is a type of substitution reaction. Halogenation reaction is a type of substitution reaction.
Q33. What is position isomerism?
Solution
When two or more compounds differ in the position of substituent atom or functional group on the carbon skeleton, they are called position isomers and the phenomenon is termed as position isomerism.
Q34. What are optically active and optically inactive compounds?
Solution
When plane polarized light is passed through a substance, it may or may not rotate the plane of the plane polarized light. The substance which does not rotate the plane of the plane polarized light is known as optically inactive compound, while a substance which rotates the plane of the plane polarized light is known as optically active substance.
Q35. How to detect halogen in an organic compound?
Solution
Halogen in an organic compound is detected by Carius method. A known mass of an organic compound is heated with fuming nitric acid in presence of silver nitrate contained in Carius tube. Carbon and hydrogen are oxidized to carbon dioxide and water. The halogen present forms silver halide, that is finally weighed.
Q36. What is crystallization process?
Solution
This is a technique for purification of solid organic compounds. It is based on the difference in the solubilities of the compound and the impurities in a suitable solvent.The impure compound is dissolved in a solvent in which it is sparingly soluble at room temperature. The solution is concentrated to get a nearly saturated solution.On cooling the solution pure compound crystsllises outand is removed by filtration.
Q37. What is reaction mechanism?
Solution
Reaction mechanism is a sequential account of each step, describing details of electron movement, energetics during bond cleavage and bond formation, and the rates of transformation of reactants into products (kinetics) is referred to as reaction mechanism.
Q38. What is lead-sulphide test for sulphur?
Solution
The sodium fusion extract is acidified with acetic acid and lead acetate is added to it. A black precipitate of lead sulphide indicates the presence of sulphur.
S2- + Pb2+ → PbS
Black
Q39. What is the Prussian blue color due to in the test for nitrogen?
Solution
In the test of nitrogen the Prussian blue color formed is of ferriferrocyanide.
Q40. What is the violet color due to, when sodium fusion extract is treated with sodium nitroprusside in the test for sulphur?
Solution
On treating sodium fusion extract with sodium nitroprusside, appearance of a violet color due to formation of [Fe(CN)5NOS]4- indicates the presence of sulphur.
Comments
Post a Comment